Multiple Choice
Identify the
letter of the choice that best completes the statement or answers the question.
|
|
| 1. | In
the following reaction,
2
Fe3+(aq) + Zn(s) ® 2 Fe2+(aq) + Zn2+(aq)
a. | Fe3+(aq) is the reducing agent and Zn(s) is the oxidizing
agent. | b. | Zn(s) is the reducing agent and Fe3+(aq) is the
oxidizing agent. | c. | Fe3+(aq) is the reducing agent and
Fe2+(aq) is the oxidizing agent. | d. | Zn(s) is the
reducing agent and Zn2+(aq) is the oxidizing agent. | e. | Zn(s) is the
reducing agent and Fe2+(aq) is the oxidizing agent. | | |
|
|
| 2. | Assuming the following reaction proceeds in the forward
direction,
Ni2+(aq) + Cr(s) ® Ni(s) +
Cr2+(aq)
a. | Ni2+(aq) is oxidized and Cr(s) is
reduced. | b. | Ni2+(aq) is oxidized and Ni(s) is
reduced. | c. | Ni(s) is oxidized and Ni2+(aq) is
reduced. | d. | Cr(s) is oxidized and Cr2+(aq) is
reduced. | e. | Cr(s) is oxidized and Ni2+(aq) is
reduced. | | |
|
|
| 3. | The
following reaction occurs spontaneously.
2 H+(aq) + Ca(s) ® Ca2+(aq) +
H2(g)
Write the balanced reduction
half-reaction. a. | 2
H+(aq) + 2 e- ® H2(g) | b. | 2
H+(aq) ® H2(g) + 2 e- | c. | H2(g)
® 2
H+(aq) + 2 e- | d. | Ca(s) + 2 e- ®
Ca2+(aq) | e. | Ca(s) ® Ca2+(aq) + 2 e- | | |
|
|
| 4. | Write
a balanced half-reaction for the reduction of ClO3-(aq) to Cl2(g) in
an acidic solution. a. | 2 ClO3-(aq) + 6 H+(aq) + 10
e- ® Cl2(g) + 6 OH-(aq) | b. | 2
ClO3-(aq) + 12 H+(aq) + 5 e- ®
Cl2(g) + 6 H2O(l) | c. | 2 ClO3-(aq) + 10 e-
®
Cl2(g) + 6 H2O(l) + 3 O2(g) | d. | 2 ClO3-(aq) + 12 H+(aq) + 10
e- ® Cl2(g) + 6 H2O(l) | e. | 2 ClO3-(aq) + 18 H+(aq)
®
Cl2(g) + 6 H3O+(aq) | | |
|
|
| 5. | Write
a balanced chemical equation for the following reaction in an acidic
solution.
Cr2O72-(aq) +
Br-(aq) ® Cr3+(aq) + Br2(aq)
a. | Cr2O72-(aq) + 2 Br-(aq) ® 2
Cr3+(aq) + Br2(aq) | b. | Cr2O72-(aq) + 6
Br-(aq) + 14 H+(aq) ® 3 Br2(aq) + 2 Cr3+(aq) + 7
H2O(l) | c. | Cr2O72-(aq) + 2
Br-(aq) + 14 H+(aq) ® 2 Cr3+(aq) + Br2(aq) + 7
H2O(l) | d. | Cr2O72-(aq) + 2
Br-(aq) + 7 H+(aq) ® 2 Cr3+(aq) + Br2(aq) + 7
OH-(aq) | e. | Cr2O72-(aq) + 6
Br-(aq) + 7 H+(aq) ® 2 Cr3+(aq) + 3 Br2(aq) + 7
OH-(aq) | | |
|
|
| 6. | All
of the following statements concerning voltaic cells are true EXCEPT a. | a salt bridge
allows cations and anions to move between the half-cells. | b. | electrons flow
from the cathode to the anode. | c. | reduction occurs at the cathode. | d. | a voltaic cell
can be used as a source of energy. | e. | a voltaic cell consists of two-half
cells. | | |
|
|
| 7. | What
is the correct cell notation for a voltaic cell based on the reaction
below?
Ni2+(aq) + Zn(s) ® Ni(s) +
Zn2+(aq)
a. | Zn(s) ï Zn2+(aq) 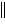 Ni2+(aq) ï Ni(s) Ni2+(aq) ï Ni(s) | b. | Zn(s) 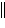 Zn2+(aq), Ni2+(aq) ï Ni(s)
Zn2+(aq), Ni2+(aq) ï Ni(s) | c. | Ni(s) 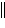 Ni2+(aq), Zn2+(aq)
Ni2+(aq), Zn2+(aq) 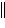 Zn(s)
Zn(s) | d. | Ni(s) ï Zn2+(aq) 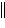 Ni2+(aq) ï Zn(s) Ni2+(aq) ï Zn(s) | e. | Ni(s) ï Ni2+(aq) 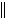 Zn2+(aq) ï Zn(s) Zn2+(aq) ï Zn(s) | | |
|
|
| 8. | Which
of the following half-reactions has been assigned a standard reduction potential,
E°, of 0.00
Volts? a. | 2
H2O(l) + 2
e- ® H2(g) + 2 OH-(aq) | b. | O2(g)
+ 4 e- ® 2 O2-(aq) | c. | Hg2Cl2(s) + 2 e- ® 2
Hg(l) + 2
Cl-(aq) | d. | Li+(aq) + e- ®
Li(s) | e. | 2 H+(aq) + 2 e- ®
H2(g) | | |
|
|
| 9. | Use
the standard reduction potentials below to determine which element or ion is the best oxidizing
agent.
Br2(l) + 2
e- ® 2 Br-(aq) E° = +1.08
V
Hg2+(aq) + e- ® Hg(l) E° = +0.86 V
Ni2+(aq) + 2 e- ® Ni(s) E° = -0.25 V
a. | Br2 | b. | Br- | c. | Hg2+ | d. | Ni2+ | e. | Ni | | |
|
|
| 10. | Which
of the following species are likely to behave as oxidizing agents: K+,
MnO4-, Cr2O72-, and
I-? a. | K+
only | b. | MnO4- and
Cr2O72- | c. | MnO4-,
Cr2O72-, and I- | d. | Cr2O72- and I- | e. | I-
only | | |
|
|
| 11. | Consider the following half-reactions:
F2(g) + 2 e- ® 2
F-(aq) E° = +2.87 V
Cu2+(aq) + 2 e- ® Cu(s) E° = +0.34 V
Sn2+(aq) + 2 e- ® Sn(s) E° = -0.14 V
Al3+(aq) + 3 e- ® Al(s) E° = -1.66 V
Na+(aq) + e- ® Na(s) E° = -2.71 V
Which of the above elements or ions will oxidize
Sn(s)? a. | Cu(s) and
Al3+(aq) | b. | F-(aq) and Cu(s) | c. | F2(g)
and Cu2+(aq) | d. | Al(s) and Na(s) | e. | Al3+(aq) and Na+(aq) | | |
|
|
| 12. | Given
the following two half-reactions, determine which overall reaction is spontaneous and calculate its
standard cell potential.
Cd2+(aq) + 2 e- ® Cd(s) E° = -1.21 V
Sn2+(aq) + 2 e- ® Sn(s) E° = -0.14 V
a. | Cd2+(aq) + Sn(s) ® Cd(s) + Sn2+(aq) 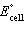 = +1.07 V = +1.07 V | b. | Cd2+(aq) + Sn(s) ® Cd(s) + Sn2+(aq) 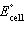 = -1.35 V = -1.35 V | c. | Cd(s) +
Sn2+(aq) ® Cd2+(aq) + Sn(s) 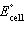 = +1.07 V = +1.07 V | d. | Cd(s) +
Sn2+(aq) ® Cd2+(aq) + Sn(s) 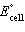 = -1.07 V = -1.07 V | e. | Cd(s) +
Sn2+(aq) ® Cd2+(aq) + Sn(s) 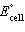 = +1.35 V = +1.35 V | | |
|
|
| 13. | Calculate 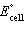 for the reaction below,
for the reaction below,
Tl+(aq) + 2 Cl-(aq) ® 2 Tl(s) + Cl2(g)
given the following standard reduction
potentials.
Cl2(g) + 2 e- ® 2
Cl-(aq) E° = +1.36 V
Tl+(aq) + e- ® Tl(s) E° = -0.34 V
a. | -2.04
V | b. | -1.70
V | c. | 1.02
V | d. | +1.70
V | e. | +2.04
V | | |
|
|
| 14. | Which
of the following equations is a correct form of the Nernst equation? a. | E =
E° - 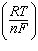 ln Q ln Q | b. | E =
E° + log 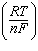 | c. | E =
E° - 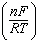 log Q log Q | d. | E =
E° - 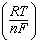 ln Q ln Q | e. | E =
E° - 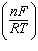 ln Q ln Q | | |
|
|
| 15. | A
Faraday, F, is defined as a. | the charge on a single electron. | b. | the charge, in
coulombs, carried by one mole of electrons. | c. | the voltage
required to reduce one mole of reactant. | d. | the current required to reduce one mole of
reactant. | e. | the charge passed by one ampere of current in one
second. | | |
|
|
| 16. | Calculate the cell potential, at 25 °C, based upon the overall reaction
3 Cu2+(aq) + 2 Al(s) ® 3 Cu(s) + 2
Al3+(aq)
if [Cu2+] = 0.75 M and [Al3+] =
0.0010 M. The standard reduction potentials are as follows:
Cu2+(aq) + e- ® Cu(s)
E° = +0.34
V
Al3+(aq) + 3 e- ® Al(s) E° = -1.66 V
a. | -2.11
V | b. | -1.26
V | c. | +1.94
V | d. | +2.06
V | e. | +2.11
V | | |
|
|
| 17. | 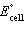 for the following galvanic cell is +0.254
V. for the following galvanic cell is +0.254
V.
Hg22+(aq) + 2 I-(aq) ® 2
Hg(l) +
I2(s)
What is DG° for this reaction? a. | -49.0
kJ | b. | -24.5
kJ | c. | +24.5
kJ | d. | +49.0
kJ | e. | +197
kJ | | |
|
|
| 18. | Calculate the equilibrium constant for the following reaction at 25 °C,
Co2+(aq) + 2 Cr2+(aq)
® Co(s) + 2
Cr3+(aq)
given the following thermodynamic
information.
Co2+(aq) + 2 e- ® Co(s)
E° = -0.28
V
Cr3+(aq) + e- ® Cr2+(aq) E° = -0.41
V
a. | 5 ´
10-24 | b. | 7 ´ 10-11 | c. | 4 ´
10-5 | d. | 2 ´ 104 | e. | 1 ´
1010 | | |
|
|
| 19. | Al3+ is reduced to Al(s) at an electrode. If a current of 2.00 ampere is
passed for 48 hours, what mass of aluminum is deposited at the electrode? Assume 100% current
efficiency. a. | 3.58
g | b. | 32.2
g | c. | 48.3
g | d. | 96.6
g | e. | 2.90
´ 102
g | | |
|